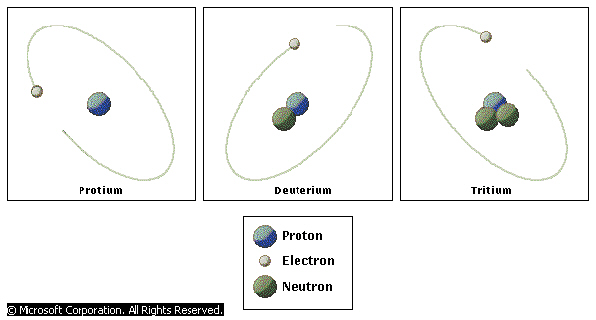

Isotopes are defined as, one of two or more species of atom having the same atomic number, hence constituting the same element, but differing in mass number. Atomic number is equivalent to the number of protons in the nucleus. Mass number is the sum total of the protons plus the neutrons in the nucleus, isotopes of the same element differ from one another only in the number of neutrons in their nuclei.
Experiments carried out early in the 20th century indicated that radioactive substances that were chemically inseparable might differ from each other only in the structure of their nuclei. The British physicist Sir Joseph Thomson demonstrated in 1912 the existence of stable isotopes by passing neon through a discharge tube and deflecting the neon ions by means of magnetic and electric fields; this showed that the stable element neon exists in more than one form. Thomson found two isotopes of neon, one of mass number 20 and another of mass 22. Later experiments showed that naturally occurring neon contains 90% of neon-20 (the isotope with mass 20), 9.73% neon-22, and 0.27% neon-21. Research on isotopes was continued by many scientists, notably the British physicist Francis William Aston; their work in detecting and studying isotopes was accelerated by the development of the mass spectrograph.
It is now known that most elements in the natural state consist of a mixture of two or more isotopes. Among the exceptions are beryllium, aluminum, phosphorus, and sodium. The chemical atomic weight of an element is the weighted average of the individual atomic weights, or mass numbers, of the isotopes. For example, chlorine, atomic wt. 35.457, is composed of chlorine-35 and chlorine-37, the former occurring with an abundance of 76% and the latter of 24%. All the isotopes of elements with atomic numbers higher than 83 (above bismuth in the periodic table) are radioactive, and a few of the lighter isotopes, such as potassium-40, are radioactive. About 280 naturally occurring stable isotopes (not radioactive) are known.