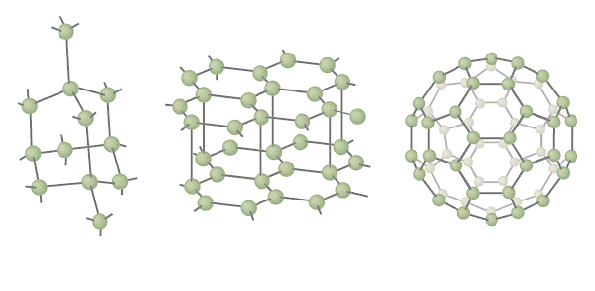
Carbon is found free in nature in three forms: amorphous, graphite, and diamond. All three are solids with extremely high melting points and are insoluble in all solvents at ordinary temperatures. Graphite is one of the softest known materials while diamond is one of the hardest. More recently, another form of carbon, buckminsterfullerene has been discovered. The molecule was named in honor of R. Buckminster Fuller, the inventor of the geodesic dome. This new form of carbon is the subject of great interest in research laboratories today.
The physical properties of the forms differ widely because of the differences in crystalline structure. In diamond, the hardest material known, each atom is linked to four other atoms in a three-dimensional framework, whereas graphite consists of weakly bonded plane layers of atoms that are arranged in hexagons. Amorphous carbon is characterized by a very low degree of crystallinity. In 1985, scientists vaporized graphite to produce a stable form of carbon molecule, buckminsterfullerene, consisting of 60 carbon atoms in a roughly spherical shape, looking like a soccer ball. The molecule may be common in interstellar dust.
Carbon, as microscopic diamonds, is found in some meteorites. Natural diamonds are found in ancient volcanic "pipes" such as found in South Africa. Diamonds are also recovered from the ocean floor off the Cape of Good Hope.
Click on the image for more information: